The objective of the analysis is the finite element analysis of different osteosyntheses on the mandible. The clinical results of different implant systems are to be analyzed and compared with each other by means of FE simulations. In this way, selected clinical findings can also be objectively substantiated by means of FE analysis.

1. Introduction
1.1 Hindered for years
Healthcare science has long sought ways to provide personalized treatments for patients. However, traditional methods for personalizing medical procedures rely heavily on experimental methodologies, which are costly and time-consuming for medical stakeholders and patients.
Since the turn of the century, physics-based simulations have become increasingly popular in engineering, thanks to recent advancements in mathematics, applied physics, and computer science.
Unfortunately, the use of physics-based simulations in medical science has been hindered by technical obstacles in digitizing and incorporating relevant data into the simulation workflow until recent years.
1.2 Precision medicine
Taking on this challenge, Simq has come to revolutionize the medical sector by enabling patient-specific simulation solutions.
As a part of the CADFEM Group founded in 2014, Simq helps all kinds of medical stakeholders to practically apply numerical simulations for more effective, personalized, and safer patient care.
Das Video wird von YouTube eingebettet und erst beim Klick auf den Play-Button geladen. Es gelten die Datenschutzerklärungen von Google,
Simq is a leading technology provider that empowers the health tech industry to reach new heights of innovation potential. By leveraging digital twin simulations and clinical applications, Simq’s cutting-edge technology platform provides easy access to physics-based computer simulations, paving the way for personalized patient treatment.
Our team of experts, with diverse backgrounds in modeling, simulation, AI, healthcare, regulation, IT, and cybersecurity, work tirelessly to enhance patient care and improve outcomes.
With Simq’s software solutions and advanced technology, complex physical and biological processes can be combined with machine learning and AI, unlocking previously untapped insights for product development, diagnosis, and therapy.
1.3 Objectives of the case study
In this case study, the objective was to use simulations as digital evidence to prove the effectiveness and safety of a medical device for post-market surveillance (PMS).
By employing finite element analysis, Simq’s advanced simulation technology allows for the analysis and comparison of different osteosynthesis techniques on the mandible.
These simulations offer medical manufacturers a cost-effective and reliable solution to provide scientific evidence to show their products’ safety, performance, and efficacy.

“With Simq and their software Simq VIT, we were able to prove the performance of our products in a scientific study in cooperation with the University Hospital Marburg and Prof. Neff. The potential of simulation in this field is for us as a manufacturer an important factor and way to secure the quality of our products in the future.”
Adrian Spiegel, Head Research/Testing, Medartis
2. Challenge: Implant Variations
Ensuring the safety and performance of medical devices is critical for any manufacturer, including continuous monitoring even after being introduced to the market. (Post Market Surveillance).
Our client Medartis is a leader in medical innovation, advancing technologies and solutions for osteosynthesis in cranial and maxillofacial (CMF) surgery and upper and lower extremities. The client actively seeks technological advancements to evaluate the quality and safety of their products.
However, when it comes to comparing different osteosynthesis on the mandible, traditional methods rely heavily on clinical data, feedback from surgeons which could be more specific and precise, or experimental procedures, which can be very time-consuming, expensive or impractical due to the indication.
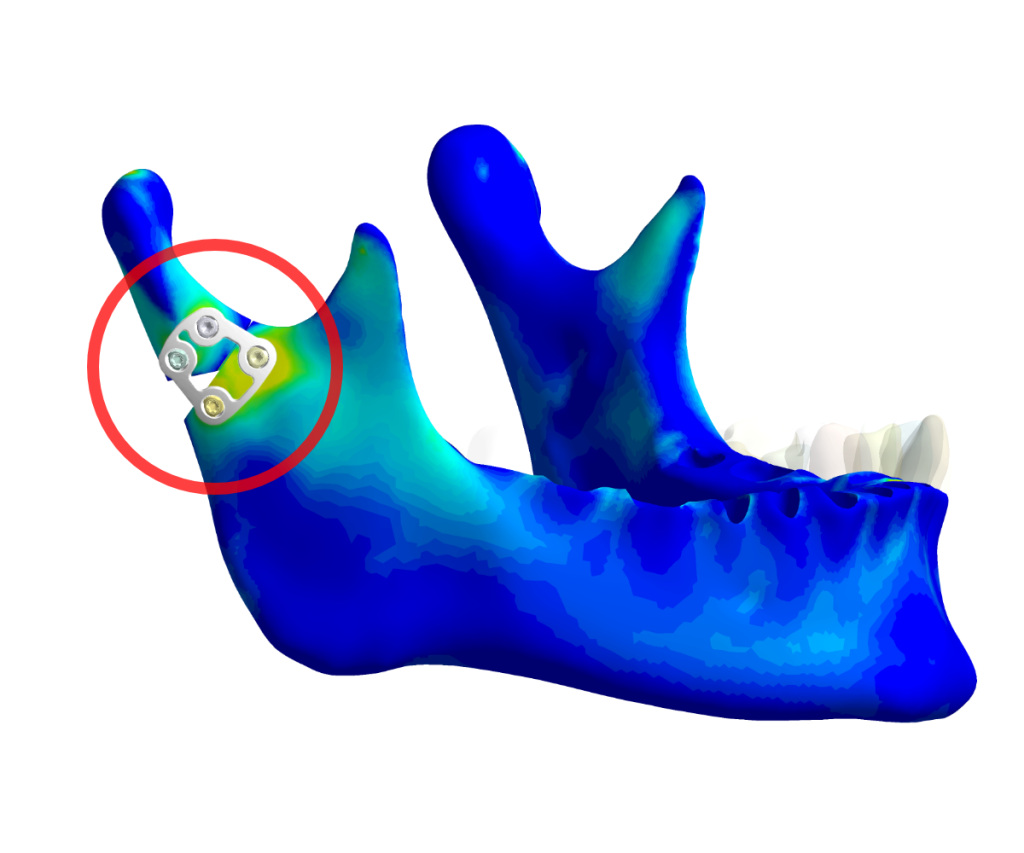
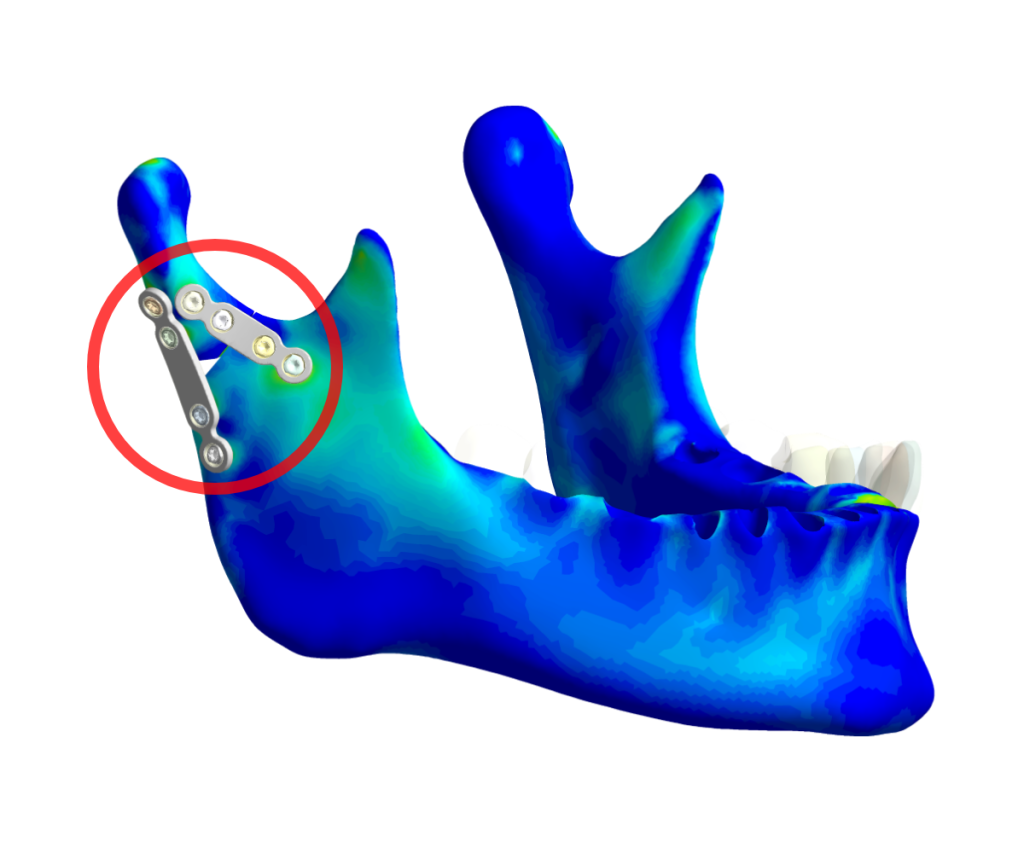
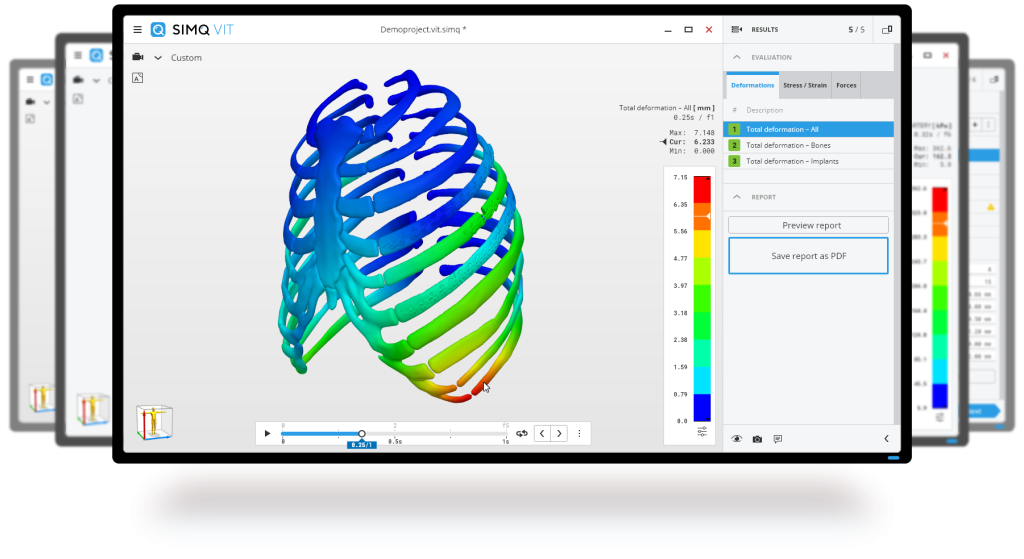
Fig. 1.: Finite element analysis of Medartis products illustrating the strain on bones for a comparative study test implant variations digitally.
3. Physics-Based Simulations To the Rescue
This is where Simq’s advanced simulation technology comes into play. With our technology platform, we can perform finite element (FE) simulations to analyze and compare different osteosynthesis on the mandible using data from an actual patient, as shown in Fig. 1.
We were able to help our client, Medartis, by simulating their existing product portfolio to compare and demonstrate the safety, efficacy, and benefits of their medical devices for post-market surveillance purposes. Our analysis utilizes physics-based simulations to compare and objectively underpin clinical data with digital evidence.
By comparing different osteosynthesis techniques on the mandible, this study demonstrates how simulations can effectively validate the procedure’s and device’s performance and ensure its safety through scientific analysis.
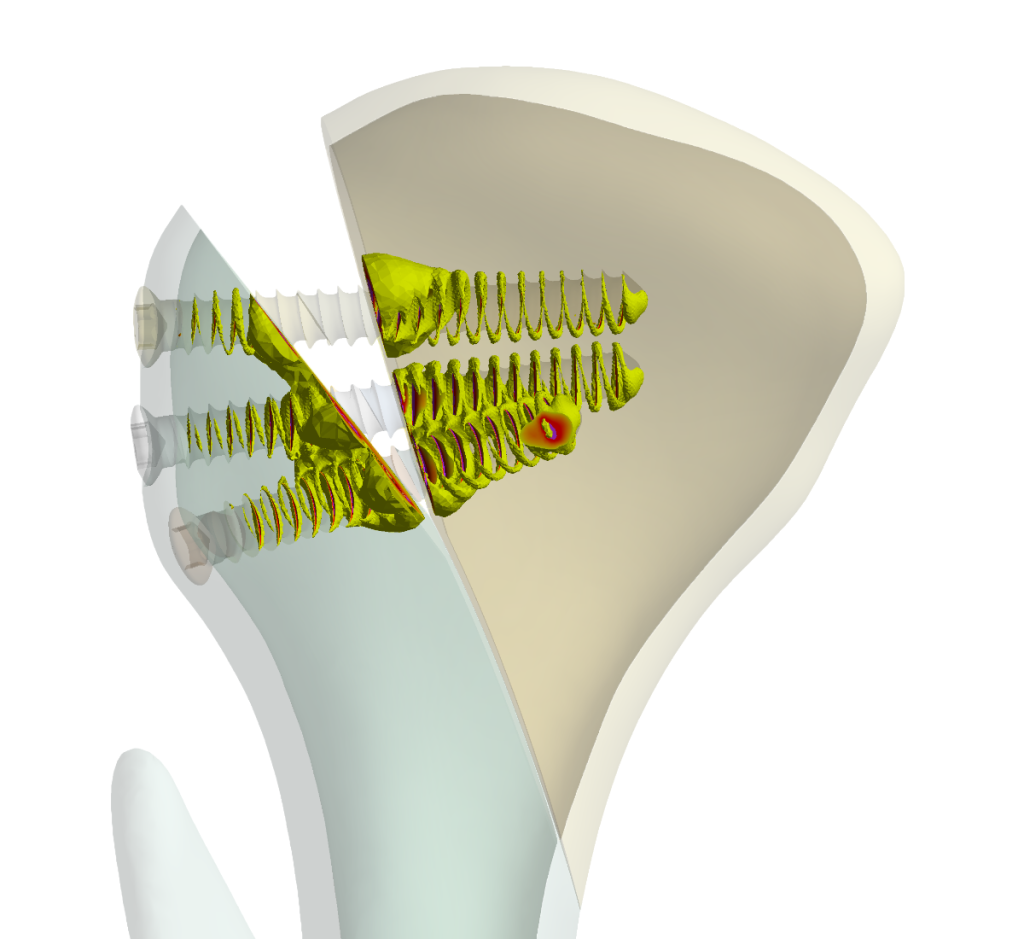
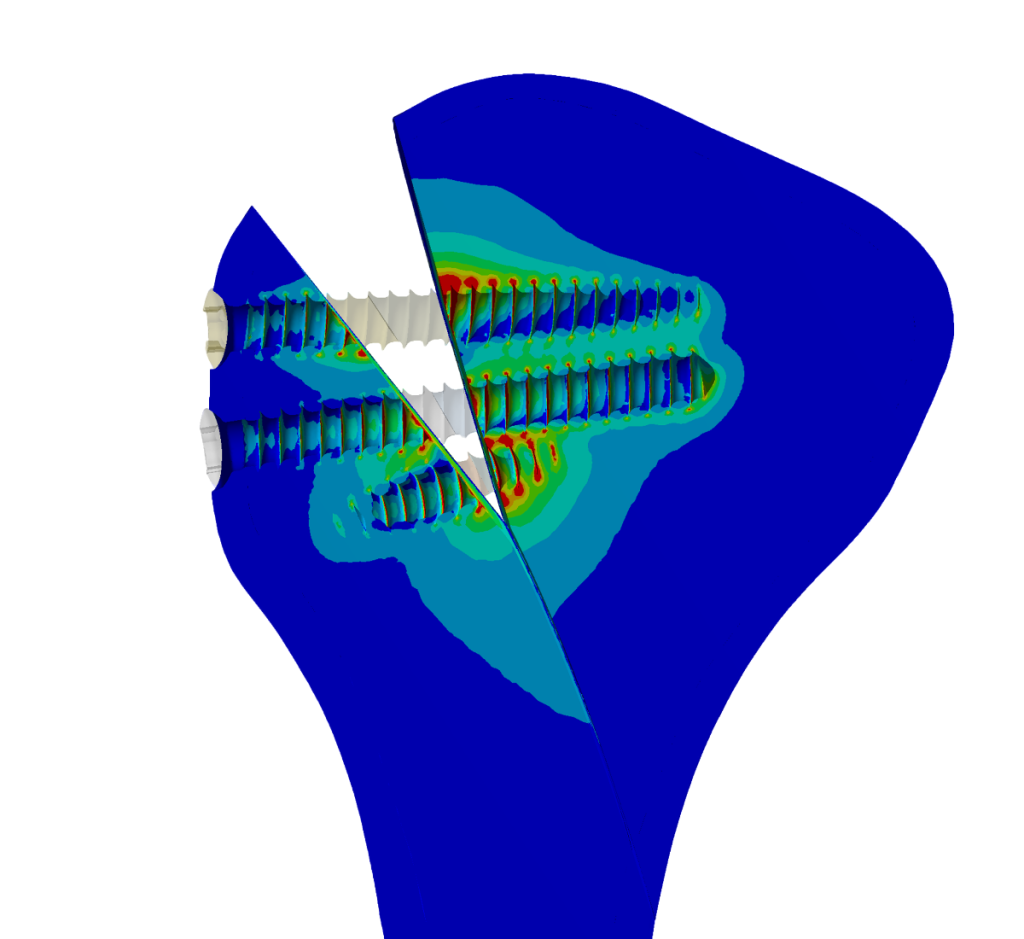
Fig. 2.: Simulation results demonstrate that using three screws in osteosynthesis provides a secure fixation on the temporomandibular joint (TMJ), as indicated by the simulation results.
Simq’s advanced simulation technology, specifically the Virtual Implant Testing (VIT) software, allows for the analysis and comparison of different osteosynthesis techniques on the mandible in a patient-specific context.
The software reproduces and substantiates clinical observations from post-market follow-up data in a virtual environment by applying physiological load boundary conditions to the implant.
These simulations serve as valuable digital evidence, supporting scientific publications and expanding technical files. Simulations play a crucial role in continuous post-market surveillance, evaluating clinical findings objectively and ensuring the device’s safety and effectiveness.
4. Results: Digital, Reliable, and Cost-Effective
The study successfully used Simq VIT to analyze and validate Medartis’ products in collaboration with Prof. Neff from the University Hospital of Marburg in Germany. The simulation results in Fig. 2 demonstrate that VIT is an effective tool for evaluating osteosynthesis implants’ clinical performance and safety during post-market surveillance.
Furthermore, the medical software allows for simulations to assess potential risks in practical use. These digital simulations provide reliable and cost-effective means to evaluate and ensure the device’s performance and safety, aligning to use simulations as digital evidence in proving the device’s effectiveness and safety.
In conclusion, using physics-based simulations as digital evidence revolutionizes the medical field by enabling personalized and cost-effective clinical treatments. These simulations complement traditional experimentation, providing valuable insights into patient-specific treatments without the limitations of time-consuming and expensive experimental procedures.
The advancements in simulation technology, exemplified by Simq’s VIT software, allow for the objective evaluation of medical devices’ performance and safety during post-market surveillance. By utilizing simulations, medical professionals can confidently provide better patient care, improve outcomes, and ensure the safety and effectiveness of medical devices.
About Simq
Simq is a regulatory platform and certified physics-based simulation software for medical diagnostics. We provide deeper insights into diagnostics and therapeutics and in-depth information for developing state-of-the-art medical devices through innovative technology.
Customers like Medartis can personalize the treatment and diagnosis using patient-specific data instead of empirical data, enabling evidence-based and patient-centered planning and decision-making before the treatment.

Simq is a certified simulation service provider and software manufacturer in the field of medicine and medical technology and is one of the pioneers of in silico medicine.





