Customer success story Patient-Specific Implant test with KLS Martin

The use of computer simulations continues to be strengthened by regulatory authorities. One goal here is to regulate mass customization in medicine more closely. FDA and MDCG want medical device manufacturers to take more responsibility for customized implants. This is where Simq VIT comes in, providing its users with fast and easy access to digital verification of patient-specific implants based on biomechanical analysis.


“With Simq VIT, we can incorporate safety-relevant information into the design of patient-specific implants and check them against the state of the art with just a few clicks. In this way, we manage the balancing act between complying with regulatory requirements and helping patients as quickly as possible.”
Thomas Kött / Head of inSilico, Division Implants / KLS Martin Group

About the customer
KLS Martin Group – “Surgical Innovation is our Passion”
The KLS Martin Group is an internationally active group of companies for innovative medical technology in almost all areas of surgery. With their innovative medical technology solutions such as implant systems, high-frequency surgical devices, surgical lasers, sterilization containers, surgical lights, surgical instruments as well as individual OR solutions, they have set new standards many times.

The task
Standard products follow a standardized, tested and documented approval process. For patient-specific implants, this has not been possible so far. To close this gap and make liability and documentation for patient-specific implants future-proof, an additional process step must be introduced.
In this process, the planning of the patient-specific implant (PSI) is to be checked and visualized in order to subsequently discuss the design together with the physician.

The solution
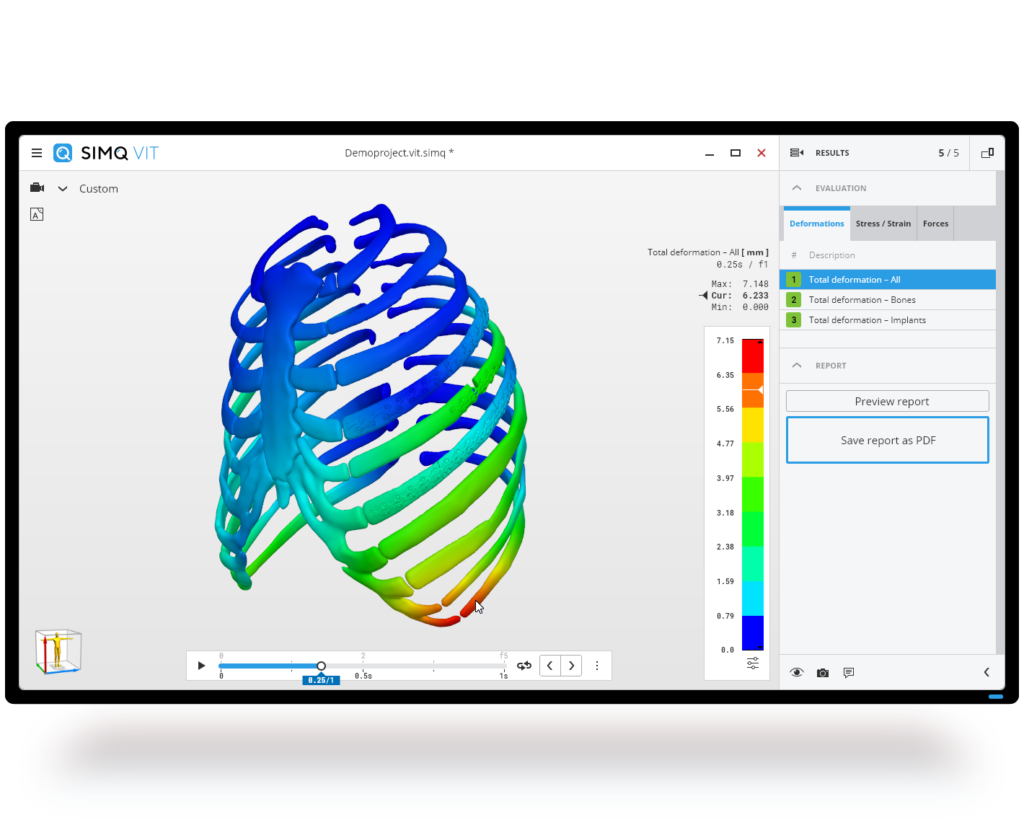
With Simq VIT, we can provide medical device manufacturers and physicians with the best possible support and digitally verify patient-specific implants quickly and easily.
The software uses finite element methods, such as those used in the automotive industry. Based on the planning data and the individual patient anatomy, an algorithm uses objective criteria to check how well the implant fits, what forces are acting and the stress levels for bone, graft and implant. Based on the simulation results, it is possible, for example, to quickly see whether all the planned screws are required and to make further improvements to the implant geometry.

The result
With Simq VIT, KLS Martin is ready for the future. Thanks to the predefined workflows in Simq VIT, which are developed together with the customer and adapted to the corresponding requirements, KLS Martin can set up highly complex biomechanical simulations within minutes and design better and patient-specific verified implants. Meanwhile, more than 100 implant designers from KLS Martin use Simq VIT on a daily basis.
Additional sources
Paper by Prof. Dr. Dr. Max Mustemann zu Mühlhausen
Written title of the paper, can also be a little longer





